Introduction:
The storage battery or secondary battery is such a battery where electrical energy can be stored as chemical energy and this chemical energy is then converted to electrical energy as and when required. The conversion of electrical energy into chemical energy by applying external electrical source is known as charging of battery. Whereas conversion of chemical energy into electrical energy for supplying the external load is known as discharging of secondary battery. The battery which uses lead and lead peroxide for the conversion of the chemical energy into electrical power, this type of battery is called a lead acid battery. The lead acid battery is most commonly used in the power stations and substations because it has higher cell voltage and lower cost. Efficiency of Lead Acid Battery is 50-95%.
Construction of Lead Acid Battery:
The Lead Acid Battery is made up of different parts:
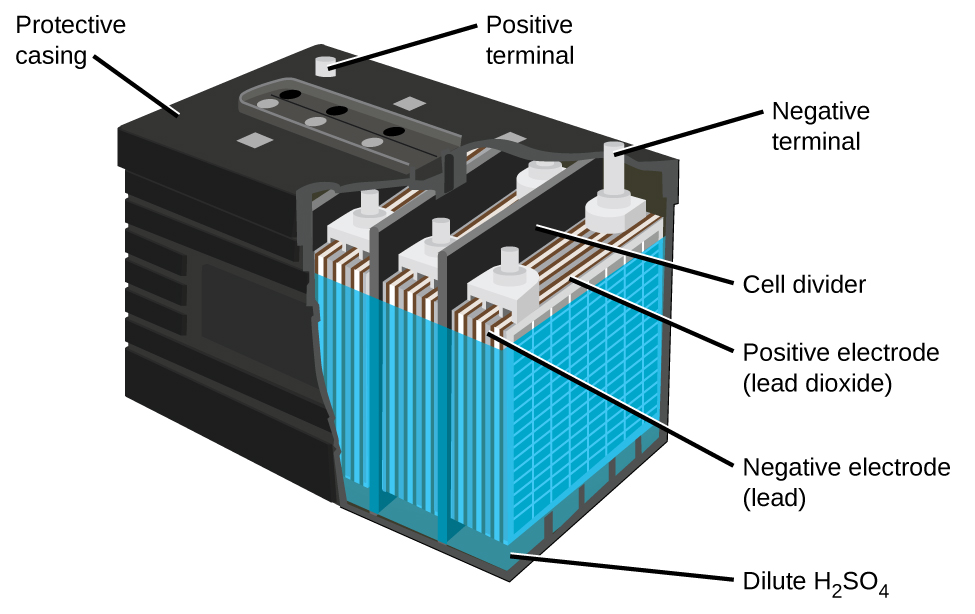
- Container– The container of the lead acid battery is made of glass, lead lined wood, ebonite, the hard rubber of bituminous compound, ceramic materials or moulded plastics and are seated at the top to avoid the discharge of electrolyte. At the bottom of the container, there are four ribs, on two of them rest the positive plate and the others support the negative plates. The material of which the battery containers are made should be resistant to sulfuric acid, should not deform or porous, or contain impurities which damage the electrolyte.
- Plates– The plate of the lead-acid cell is made up of lead and active material. they all form some type of grid, it is essential for conducting the electric current and for distributing the current equally on the active material. The grids are made up of an alloy of lead and antimony. The plates of the battery are of two types. They are the formed plates or plante plates and pasted or faure plates.
- Active Material– The material in a cell which takes active participation in a chemical reaction (i.e. absorption of electrical energy) during charging or discharging is called the active material of the cell. The active elements of the lead acid are:
- Lead peroxide (PbO2) – It forms the positive active material. The PbO2 are dark chocolate broom in colour.
- Lead – Its form the negative active material. It is grey in colour.
- Dilute Sulfuric Acid (H2SO4) – It is used as an electrolyte. It contains 31% of sulfuric acid.
- Separators– The separators are thin sheets of non-conducting material made up of chemically treated leadwood, porous rubbers, or mats of glass fibre and are placed between the positive and negative to insulate them from each other.
- Battery Terminals– A battery has two terminals the positive and the negative.
Working of Lead Acid Battery:
During charging of battery, current is passed through it which causes some chemical changes inside the battery. This chemical changes absorb energy during their formation. When the battery is connected to the external load, the chemical changes take place in reverse direction, during which the absorbed energy is released as electrical energy and supplied to the load.
The lead acid storage battery is formed by dipping lead peroxide plate and sponge lead plate in dilute sulfuric acid. A load is connected externally between these plates. In diluted sulfuric acid the molecules of the acid split into positive hydrogen ions (H+) and negative sulfate ions (SO4 2-). The hydrogen ions when reach at PbO2 plate, they receive electrons from it and become hydrogen atom which again attack PbO2 and form PbO and H2O (water). This PbO reacts with H2 SO4 and forms PbSO4 and H2O (water).

SO4 2- ions are moving freely in the solution so when they will reach to pure Pb plate where they give their extra electrons and become radical SO4. As the radical SO4 cannot exist alone it will attack Pb and will form PbSO4. As H+ ions take electrons from PbO2 plate and SO4 2- ions give electrons to Pb plate, there would be an inequality of electrons between these two plates. Hence there would be a flow of current through the external load between these plates for balancing this inequality of electrons. This process is called discharging of lead acid battery. During discharging,
- Both of the plates are covered with PbSO4.
- Specific gravity of sulfuric acid solution falls due to formation of water during reaction at PbO2 plate.
- As a result, the rate of reaction falls which implies potential difference between the plates decreases during discharging process.
Now, when we connect an external DC source to the termial, Hydrogen ions (cation) being positively charged, move to the electrode (cathode) connected with negative terminal of the DC source. Here each H+ ion takes one electron from that and becomes hydrogen atom. These hydrogen atoms then attack PbSO4 and form lead and sulfuric acid.

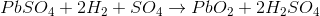
SO42- ions (anions) move towards the electrode (anode) connected with positive terminal of DC source where they will give up their extra electrons and become radical SO4. This radical SO4 cannot exist alone hence reacts with PbSO4 of anode and forms lead peroxide (PbO2) and sulfuric acid (H2SO4). So, during charging,
- Lead sulfate anode gets converted into lead peroxide.
- Lead sulfate of cathode is converted to pure lead.
- Terminal potential of the cell increases.

You must be logged in to post a comment.